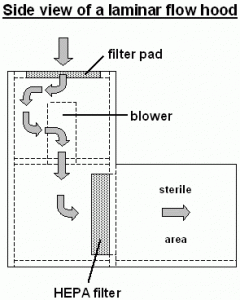
LAF : Laminar Air Flow – Airflow which is linear and positive up to working surfaces and thus prevents contamination of surrounding viable/ non viable particulate matter in aseptic handling. The Operation, Cleaning, Maintenance and Qualification of Laminar Air Flow (LAF) shall discussed in below SOP.
Laminar Air flow (LAF) – Operation, Cleaning and Qualification
Purpose:
- To lay down the procedure for the operation, cleaning, maintenance and qualification of the Laminar Air Flow.
Scope:
- This Standard Operating Procedure is applicable at Microbiology section / Sterile Area where Laminar Air Flow (LAF) installed.
References & Annexures:
-
References:
- Operation Manual of Laminar Air Flow (LAF) for Operation, Performance check, Sanitization and Qualification.
-
Annexures:
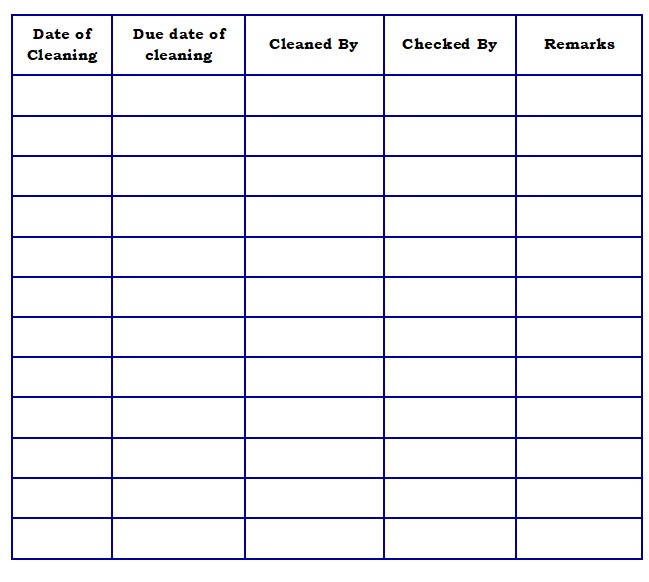
- Annexure-1 : Cleaning Record of Prefilter
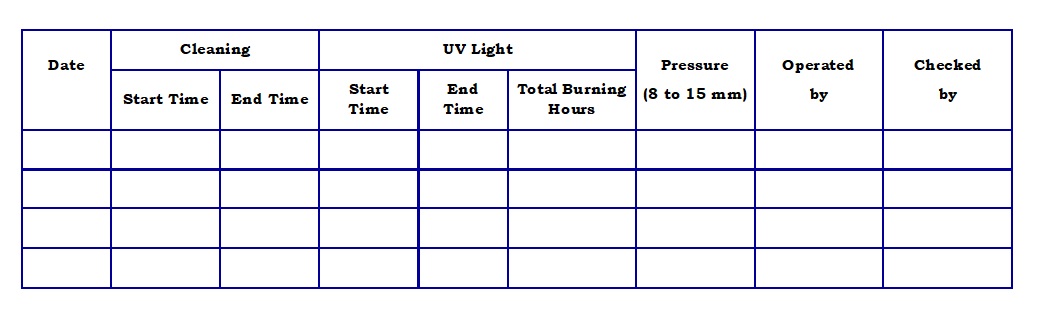
- Annexure-2 : Laminar Air Flow operation Record.
- Annexure-3 : Qualification Status Label
Responsibilities:
-
Microbiologist / Executive – Quality control.
-
Quality Assurance (QA) Department:
- To review the SOP.
- To ensure the implementation of SOP.
-
Engineering and Maintenance Department:
- Review the SOP.
- Perform the activity as per procedure provided.
- Ensure the implementation of SOP.
-
Regulatory Affairs, Quality Head and Plant Head :
- To review and approve new or revised SOPs.
Distribution:
- Quality Control Department
- Quality Assurance Department
- Engineering and Maintenance Department
Abbreviations and Definition of Terms :
-
Abbreviations :
- CC : Change Control
- °C : Degree Celsius
- HEPA : High Efficiency Particulate Air
- IPA : Iso Propyl Alcohol
- LAF : Laminar Air Flow
- NA : Not Applicable
- PAO : Poly Alfa Olefin
- QA : Quality Assurance
- QC : Quality Control
- SOP : Standard Operating Procedure
- UV : Ultra Violet
-
Definition of Terms :
- Standard Operating Procedure (SOP): A written authorized procedure, which gives instructions for performing operations.
- LAF: Laminar Air Flow: Airflow which is linear and positive up to working surfaces and thus prevents contamination of surrounding viable/ non viable particulate matter in aseptic handling.
Procedure (Laminar Air Flow – LAF):
-
Cleaning of Laminar Air Flow (LAF)
- First of all, Turn OFF the switch of the Air Flow and UV of LAF unit (if ON).
- Then clean properly the working chamber from ceiling, followed by side glass and then the platform of the chamber with 70% IPA.
- After that spray 70% IPA and switch off the visible light and then switch ON the UV light till to start the next operation.
- Finally Routine culture handling shall perform at the end of the Sample testing only.
-
Laminar Air Flow Operation (LAF-Operation)
- Switch ON the mains.
- Ensure that the manometer is showing Zero reading before starting.
- Turn the switch of the Air Flow to ON position. Ensure that the differential pressure is within the limit of 8 to 15 mm of water and record the observation in Annexure-2.
- Turn the switch of the UV lights to ON position.
- After 30 minutes switch OFF the UV and switch ON the Visible light. Enter the burning hour details of UV in Annexure-2.
- Now perform the activities.
Note: Replace the UV Light after 999 Burning hrs.
-
Pre-Filter Cleaning of Laminar Air Flow (LAF) :
- First of all, Switch ‘OFF’ the mains of LAF, loosen the bolts properly and take out the Prefilter slowly, without any jerks from the LAF.
- Then Shift the filter to wash area outside microbiology analysis room where cleaning is to be done.
- Ensure the left out opening of the Prefilter on the LAF unit shall covered with butter paper in order to prevent dust as well as the outside air moving inside the LAF unit.
- First de-dust the filter properly with the help of dry, oil free compressed air. The compressed air shall be forced properly to prefilter of LAF otherwise it will damage the filter.
- After dedusting, clean the prefilter properly 2 -3 times with Purified water. Allow all the water to drain completely after water cleaning.
- Then dry the prefilter after proper draining in a clean dry environment free from the dust; otherwise the chances of contamination in the sterile environment may arise or it may decrease the efficiency of the prefilter. Do not dry the prefilter in an open environment.
- Spray 70% IPA solution filtered through 0.45 m filter and mop SS attachment of the prefilter properly before transferring inside the microbiology analysis area.
- Affix the cleaned prefilter properly in appropriate place by removing the butter paper cover and tighten the bolts properly.
-
Check the Differential Pressure in Laminar Air Flow (LAF) :
- Then Place the prefilter at its position. Switch on the Laminar Air Flow (LAF) unit. Check the differential pressure is in between 8 to 15 mm of water gauge.
- If pressure reading on manometer is not achieved, put a status label ‘Under Maintenance’ & intimate the Maintenance Department for necessary action.
- Prefilter of the Laminar Air Flow (LAF) benches shall clean once in three months In order to retain the efficiency of the HEPA filter and proper working of LAF.
- After the Prefilter is fixed, the area shall clean properly followed by fogging as per SOP of “Fumigation of Microbiology” before using the instrument as well as the area for operation.
- Cleaning record is to maintain as per Annexure-1.
- Clean the Pre filter at the frequency of 3 months ± 7 days.
Also visit : Cleaning and Sanitization in Pharmaceuticals
-
Qualification of Laminar Air Flow (LAF) :
-
Frequency:
- Once in six months ±15 days.
-
The qualification of LAF required:
- If any failure observed during routine monitoring.
- In case of breakdown, maintenance, preventive maintenance.
- In case of change in installation location.
-
Procedure of LAF Qualification :
- Following tests are performed during qualification and results are within acceptance criteria.
-
Air velocity test:
- Ensure that air velocity of the LAF is checked using calibrated anemometer.
- Acceptance criteria: The velocity of the laminar air should be within 90 + 20 % ft/min.
-
Particulate count test :
- Ensure that particle count of the work station is done using a calibrated air borne particulate matter counter. 1 m3 of air sample is collected per filter.
-
- Following tests are performed during qualification and results are within acceptance criteria.
- Acceptance criteria:
- ≥ 0.5µ particles: NMT 3500 particles per m3 ≥ 5.0µ particles : 00 (Nil / Absent) particles per m3
-
-
HEPA Filter integrity test / HEPA Filter leak test (Laminar Air Flow – LAF):
-
- Ensure that the filter integrity test of the HEPA filter done using PAO (Poly Alfa Olefin).
- Acceptance criteria:
- The penetration level should less than 0.01%.
- In case of detection of rupture of fine filter, seal the rupture with soft silicone sealant.
- Restrict the silicone sealing to not more than 3.0% of the area of the filter and not more than 3.0 sq. inch of single patch.
- Qualification shall perform by external agency if facility not available at location.
- After Completion of Qualification write the details and pest as per Annexure-3.
For basics of Laminar air flow Click here
Cleaning Record of Prefilter – LAF
Laminar Air Flow Operation Record
Visiters also reading :
A) Isolation and Identification of Microorganisms
B) Laminar Airflow (LAF) – Operation, Cleaning, and Qualification




Pingback: How Does A Laminar Air Flow Work - Tech Hangouts
Pingback: Microbial Culture Management SOP - Pharma Beginners